Faecal Calprotectin Assay Range
Faecal Calprotectin Test– Your Choice
The widest range of calprotectin stool test formats for assessment within primary care, Gastroenterology clinics, the hospital laboratory and for patient self testing.
Faecal calprotectin is a reliable, proven protein biomarker that enables non-invasive differentiation between patients with Irritable Bowel Syndrome (IBS) and those with Inflammatory Bowel Disease (IBD). The simple diagnostic test is helping to improve patient care and reduce health care costs by screening out IBS patients and preventing the need for unnecessary endoscopy procedures.
It is also proving helpful in monitoring known IBD patients to predict potential relapse and adjust medication accordingly.
Alpha Laboratories is the exclusive UK partner for BÜHLMANN Calprotectin, supporting the full range of calprotectin assay kit formats, and has many years expertise in this area.
Supporting New Standards of Care for IBD with the first CE marked Calprotectin Patient Self-Test
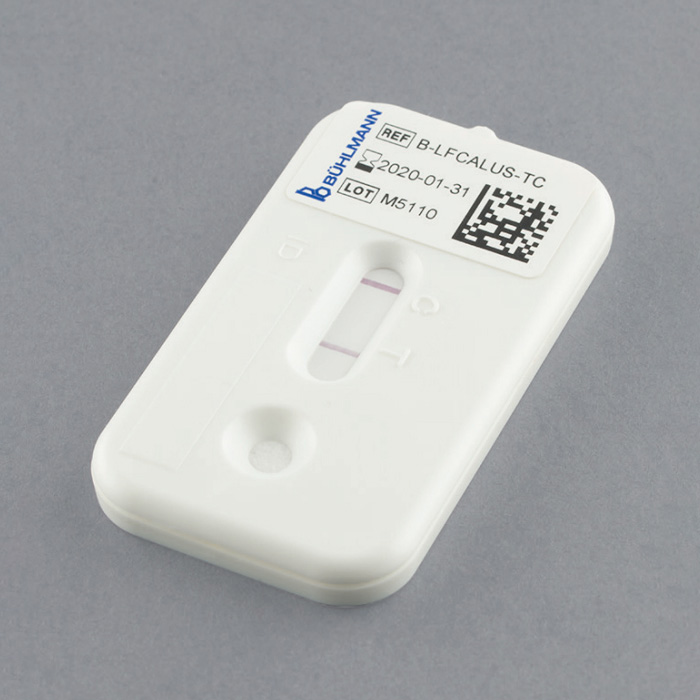
A rapid quantitative Faecal Calprotectin testing platform for fast turn around, lower throughput and Point-of-Care requirements.
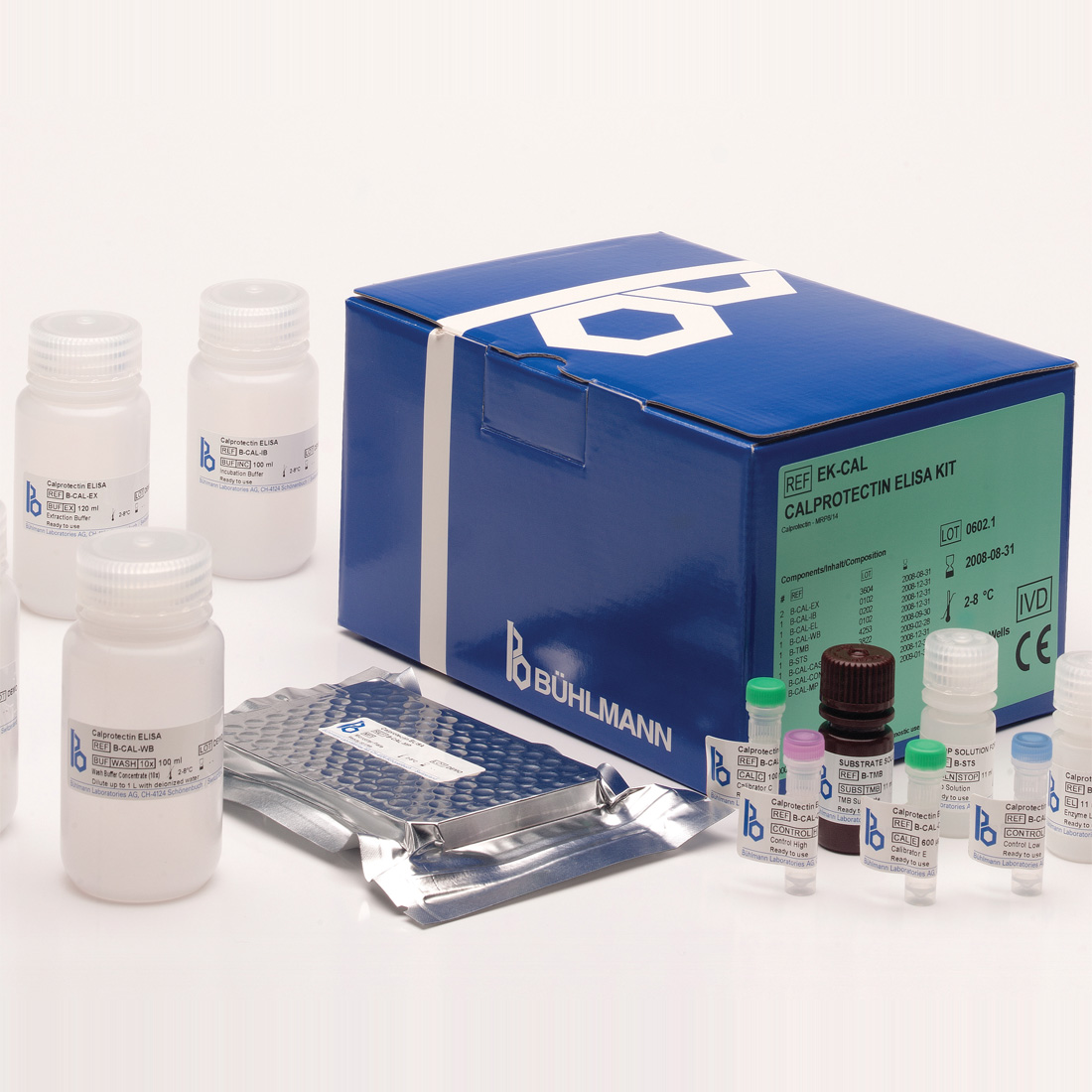
The BÜHLMANN fCAL® ELISA (EK-CAL) is the UK market-leading calprotectin ELISA.
It provides quantitative in vitro determination of faecal calprotectin in high sample throughput environments.
Speed, Quality and Flexibility in Calprotectin Quantification
BÜHLMANN fCAL® turbo is a flexible turbidimetric assay for faecal calprotectin applicable on many major clinical chemistry platforms.
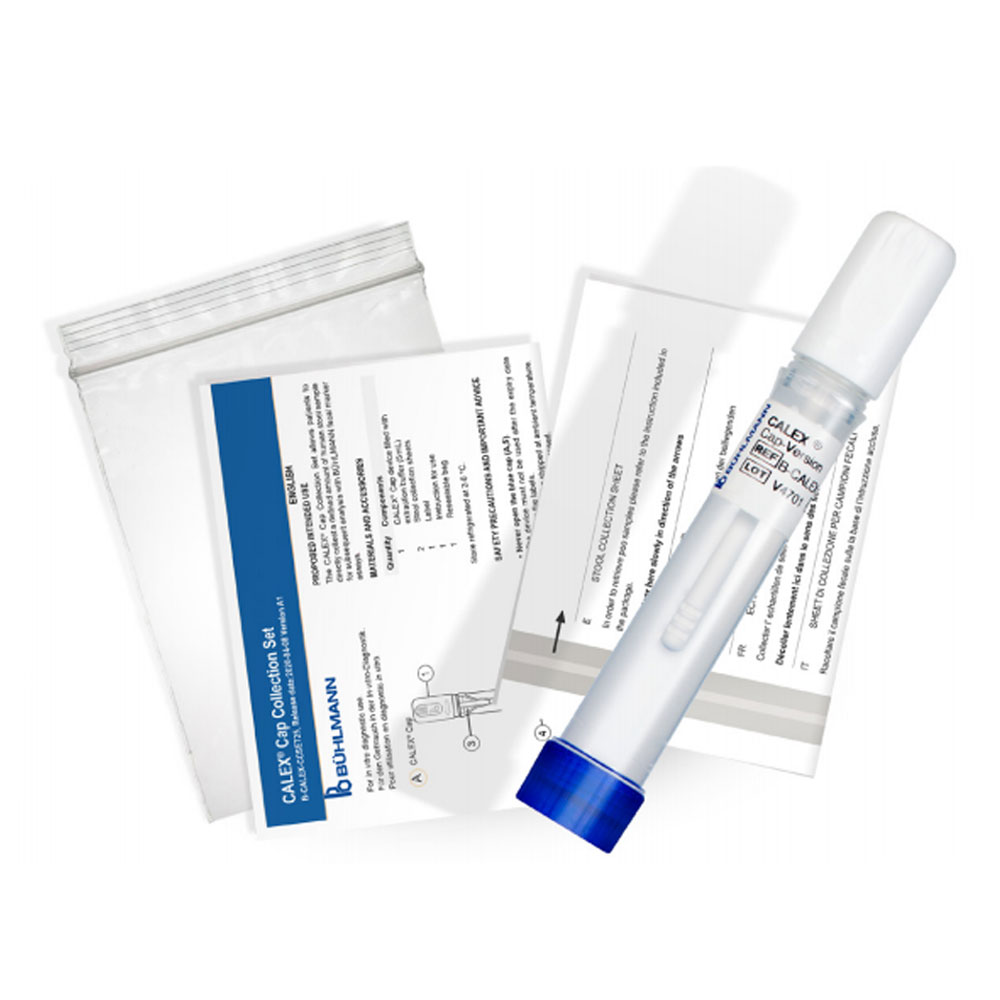
Dedicated, specially designed devices to minimise operator contact with the sample, whilst optimising extraction of calprotectin, to simplify and assist the pre-analytical faecal extraction process.
CALEX patient packs enable the patient to directly prepare the CALEX themselves. It can then be sent to the laboratory, rather than supplying a stool sample in a container.